The Next Phase of GLP-1s Has Arrived

I believe we will look back at history as B-GLP and A-GLP — Before and After. Their impact on the human race is that significant.
If nothing goes wrong long-term, they will eventually be as widely used as statins.
But there has always been one problem.
They don’t fit into real life.

For the past few years, we’ve been asking patients to do something unusual:
Inject themselves once a week.
Store their medication in the fridge.
Fight their insurance just to get access.
Even the recently released pill version of GLP-1s—Novo Nordisk’s oral Wegovy—comes with its own ritual:
Wake up early.
Take it on an empty stomach.
Drink a few ounces of water.
Wait 30 minutes before doing anything else.
That’s not a medication.
That’s a routine.
And routines don’t scale.

Last week, the FDA approved a new weight loss drug from Eli Lilly called Foundayo (orforglipron).
In my opinion, this is the most important GLP-1 development since Ozempic.
Not because it’s more effective.
But because it’s finally usable for the masses.
Ozempic proved GLP-1s work.
Foundayo is how they scale.
I’ve been tracking this approval for months. I even wrote about it in detail back in August. Now that it’s finally here, let’s break it down even further…
Let’s start with the size of the opportunity.
The CDC estimates that over 40% of American adults—more than 100 million people—have obesity.
And yet, even after years of Ozempic mania, only a fraction of that population is actually taking GLP-1 drugs. A recent KFF poll found that about 12% of U.S. adults report currently using a GLP-1.
We have a drug that works.
We have a massive eligible population.
And we’re still nowhere close to saturation.
Why?
Because the biggest barrier to GLP-1 adoption was never science.
It was friction.

Foundayo is the first oral GLP-1 that actually behaves like a normal medication.
Unlike oral Wegovy, you can take it:
at any time of day
with or without food
with any beverage
without waiting 30 minutes to live your life
No rituals. No constraints. No friction.
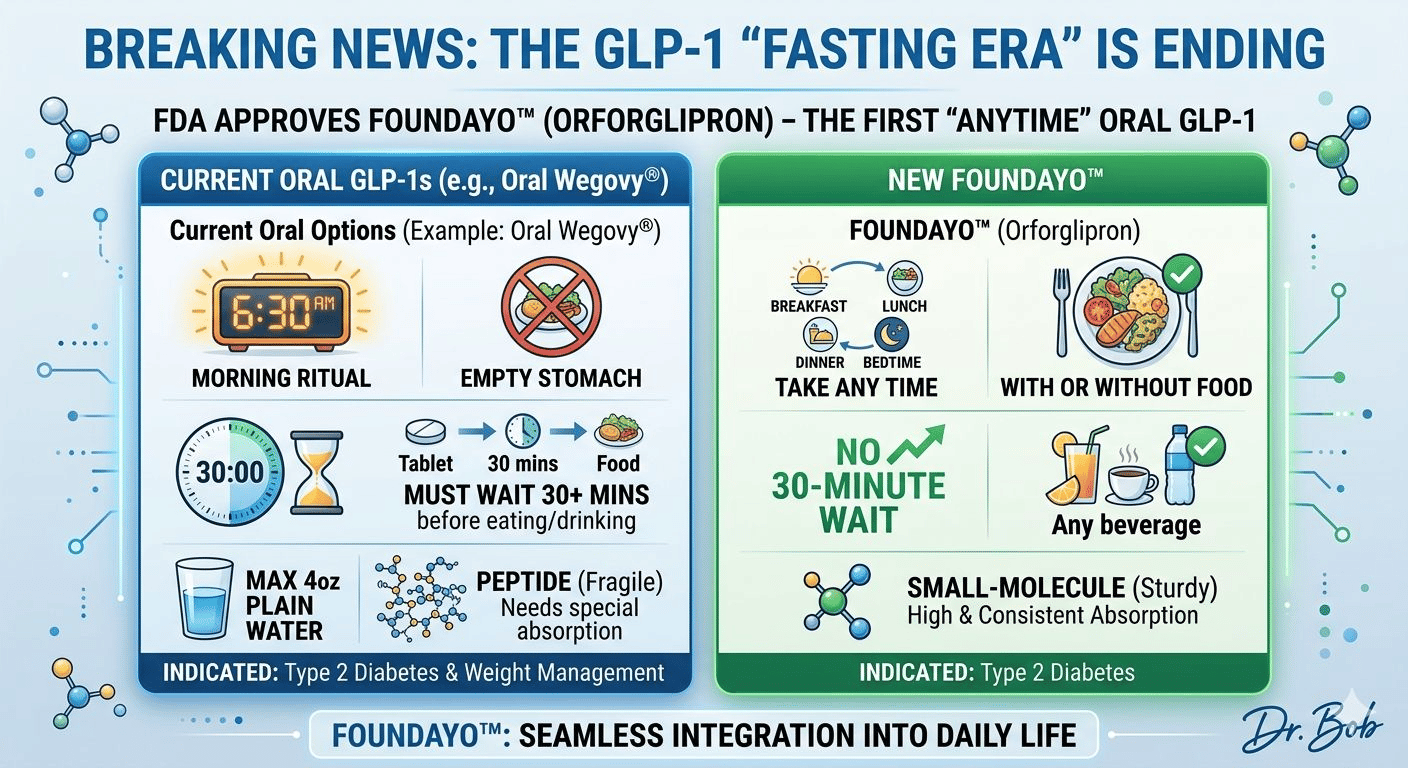
I stole this from a Harvard medical professor on LinkedIn named Dr. Bob
This is the first GLP-1 designed for real life, not clinical trials.
Sure, if you look at the data, Foundayo is slightly less effective than Novo’s Wegovy pill.
“Novo Nordisk’s Wegovy pill helped people lose about 16.6% of their body weight on average when taken for 64 weeks in a trial. For Lilly’s pill, it was an average reduction of 12.4% after 72 weeks.”
That will be the headline in a lot of places.
It’s also the wrong takeaway.
The entire conversation about “which GLP-1 is more effective” is about to become irrelevant.
Because Foundayo clears a much more important threshold:
It is effective enough to justify mass adoption.
Once “good enough” is achieved → usability dominates.

Think about statins (aka cholesterol meds).
They are not perfect drugs.
They don’t work equally well for everyone.
They come with side effects.
But they are:
easy
cheap
widely available
have inarguable benefit for all
So tens of millions of people use them.
That’s what scale looks like.
GLP-1s have never truly had that.
Until now.

The Real Story Isn’t Switching. It’s Starting.
A lot of people are going to look at Foundayo and think:
“People will switch from injections to pills.”
And sure, that will happen.
But that’s not the story.
The story is the millions of people who never started in the first place.
GLP-1s don’t scale by converting users. They scale by removing excuses.
We already have proof that making GLP-1s easier to use brings in new patients.
Novo’s Wegovy pill hit 94,000 U.S. prescriptions in a single week just 11 weeks after launch—and many of those prescriptions were written for people who had never used a GLP-1 injection before.
That’s the signal.
There are still tens of millions of people who would benefit from GLP-1s who are sitting on the sidelines because of:
injections
inconvenience
or the simple psychological barrier of feeling like the treatment is “too serious”
A pill changes that.
Not clinically.
Psychologically.
It makes the treatment feel normal.
And once something feels normal, it spreads.

Speed Matters More Than You Think
There’s another part of this story that isn’t getting enough attention (and is extremely timely given what I wrote about last week!).
The FDA approved Foundayo at record speed—one of the fastest approvals of its kind in over two decades. It was the first “New Molecular Entity” approved under the “Commissioner’s National Priority Voucher Program”.
It was approved only 50 days after filing - which is 294 days before expected.
That’s a big deal.
Because speed isn’t just about getting drugs to market faster.
It’s about giving companies the room to actually lower prices.
Every extra month of exclusivity on the market matters.
It gives companies more time to:
generate revenue
expand access
lock in market share before competition shows up
And in a market like GLP-1s—where demand is already overwhelming—that time is incredibly valuable.

The Pricing Signal
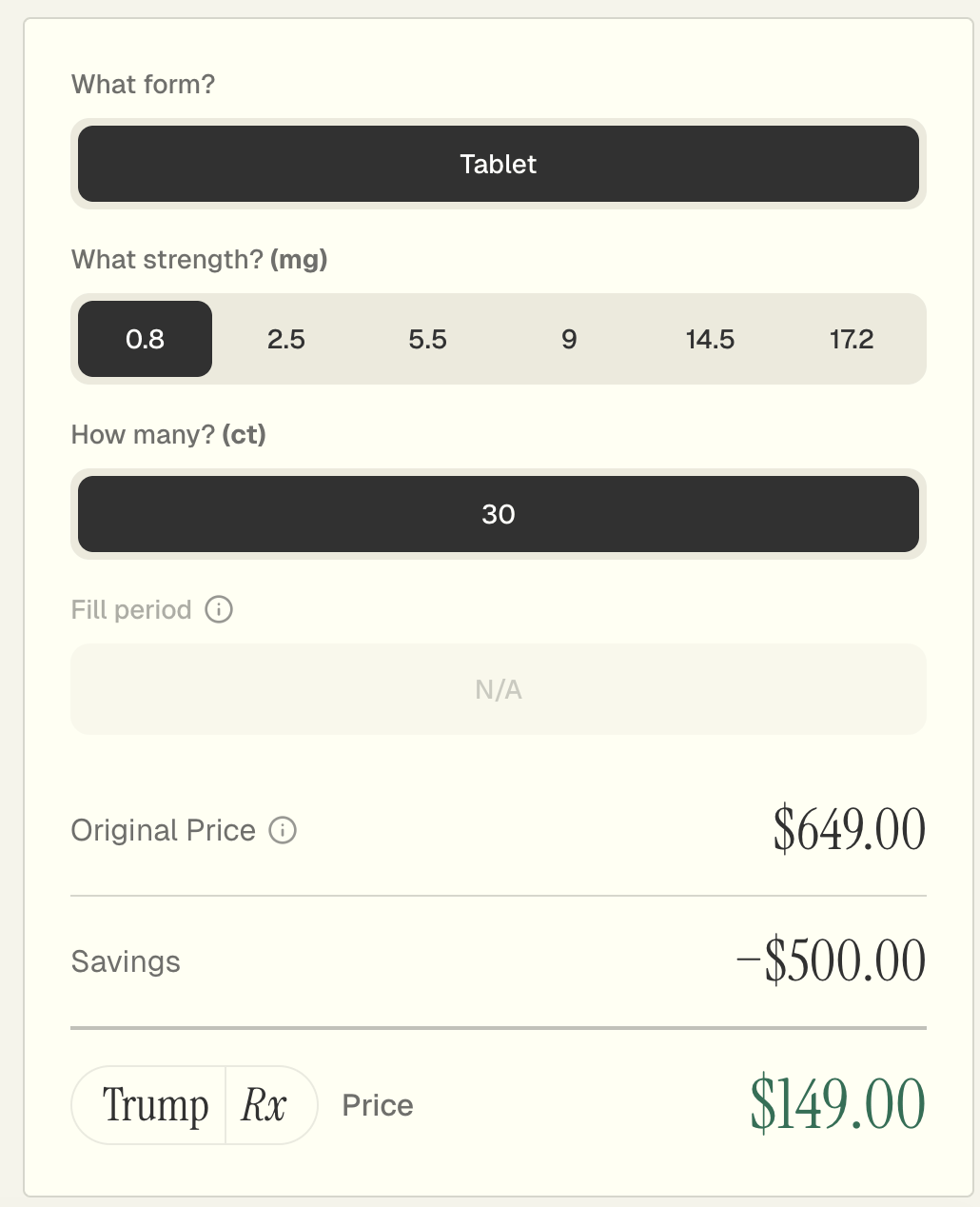
Even though Foundayo is not yet available to pharmacies…

This is what I see when I try to order Foundayo for my pharmacy, C.O. Bigelow.
…a bit of detective work (shoutout to Brian Reid) shows that TrumpRx currently lists Foundayo at a $649 monthly list price, with direct-to-consumer patients paying around $149 per month.
Let’s be clear.
That’s not cheap.
But in the context of GLP-1s… it kind of is.
We are talking about a class of drugs that has routinely cost:
$1,000+ per month
required refrigeration
required injections
and still managed to explode in demand
Now you have:
a pill
no storage constraints
no administration friction
and a meaningfully lower price point
I personally believe that many new patients will be able to justify that monthly price with how much they will save on the food they won’t be buying once they start on the drug.
And not that anyone cares about my wallet, but a list price around 50% of what I currently pay for a one-month supply of Mounjaro will work wonders for my pharmacy’s cash flow!

Wall Street seems to understand where this is going.
Analysts are already projecting that Foundayo could generate around $21 billion in global sales by 2030, compared to roughly $4 billion for Novo’s Wegovy pill.
I repeat - that’s not a bet on superior efficacy.
That’s a bet on scale.

Zoom Out
We’ve spent the last few years proving that GLP-1s work.
That was Phase One.
This is Phase Two.
This is where:
the drugs get simpler
the access gets wider
the pricing starts to move
and the patient base expands dramatically
Ozempic was the breakthrough.
Foundayo is the rollout.
Three years ago, nobody knew what a GLP-1 was.
Three years from now, it will be unusual not to be on one.
Giddy up.

Alec Wade Ginsberg, PharmD, RPh
4th-Gen Pharmacist | Owner & COO, C.O. Bigelow
Founder, Drugstore Cowboy